Under these circumstances, no electron could lose energy because no electron could move down to a lower energy level. Bohr model or RutherfordBohr diagram, presented by Niels Bohr and Ernest Rutherford in 1913. Bohr worked out rules for the maximum number of electrons that could be in each energy level in his model, and required that an atom in its normal state (ground state) had all electrons in the lowest energy levels available. In this situation, no person could move down because all of the lower rungs are full. In 1913, after returning to Copenhagen, he began publishing his theory of the simplest atom. Bohr, shown in Figure 22.8, became convinced of its validity and spent part of 1912 at Rutherford’s laboratory. If the ladder had five people on it, they would be on the lowest five rungs. Niels Bohr (18851962), a Danish physicist, did just that, by making immediate use of Rutherford’s planetary model of the atom. Other rules for the ladder are that only one person can be on a rung in the normal state, and the ladder occupants must be on the lowest rung available. Suppose we had such a ladder with 10 rungs. The only way you can be on that ladder is to be on one of the rungs, and the only way you could move up or down would be to move to one of the other rungs. Appendix I Review of Basic Chemistry A-3. 
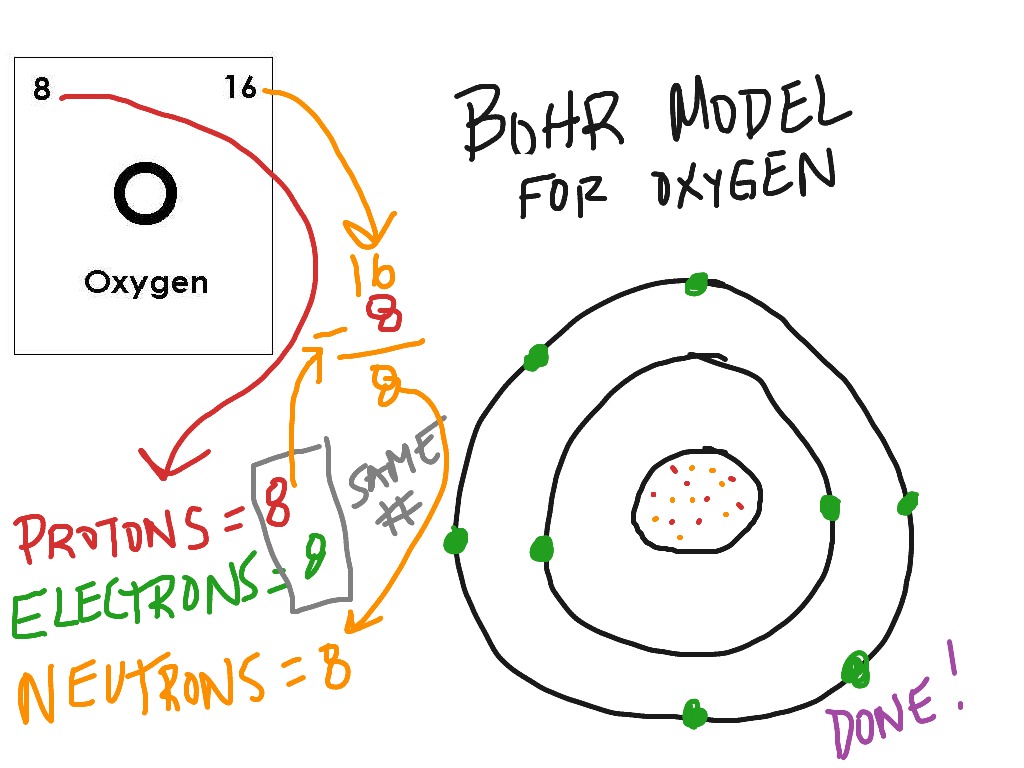
Oxygen (O) Water molecule (H2O) Figure A.4. Start by sketching the nucleus, and then draw the two electron shells. In this drawing, known as a Bohr model, electrons move in orbitals. Note that Bohr stated that electrons in the atom follow elliptical orbits (not. To draw the fluorine Bohr model, note the 9 protons, 10 neutrons, and 9 electrons. In 1913, Danish physicist Niels Bohr applied Max Planck’s quantum theory to the nuclear atom of Ernest Rutherford, thus formulating the well-known planetary model of the atom, wherein electrons orbit a central nucleus in well-defined levels of energy ( Figure 1 ). Encircling this nucleus are two electron shells, carrying a total of 9 electrons. It would be like a ladder that had rungs only at certain heights. Download this Premium Vector about Oxygen atom bohr model, and discover more than 143 Million Professional Graphic Resources on Freepik. In the fluorine Bohr model, the nucleus holds 9 protons and 10 neutrons. (b) The energy of the orbit becomes increasingly less negative with increasing n. 2 The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. Figure 2 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. In this state the radius of the orbit is also infinite. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. The energy levels are quantized, meaning that only specific amounts are possible. Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. Note that the spacing between rungs gets smaller at higher energies (CC BY-NC Ümit Kaya)īohr hypothesized that the only way electrons could gain or lose energy would be to move from one energy level to another, thus gaining or losing precise amounts of energy. \): The energy levels of the electrons can be viewed as rungs on a ladder.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
